- Home
- -
- Breast-SlimView®
The patented clinical decision support software for 2D and 3D mammography: Breast-SlimView®
Hera-MI has developed Breast-SlimView®, a patented clinical decision support system for 2D and 3D mammography. Breast-SlimView® offers an innovative and disruptive reading support where only relevant information is displayed. Radiologists will be able to free themselves from overloaded information and visualize potentially suspicious areas at a glance.
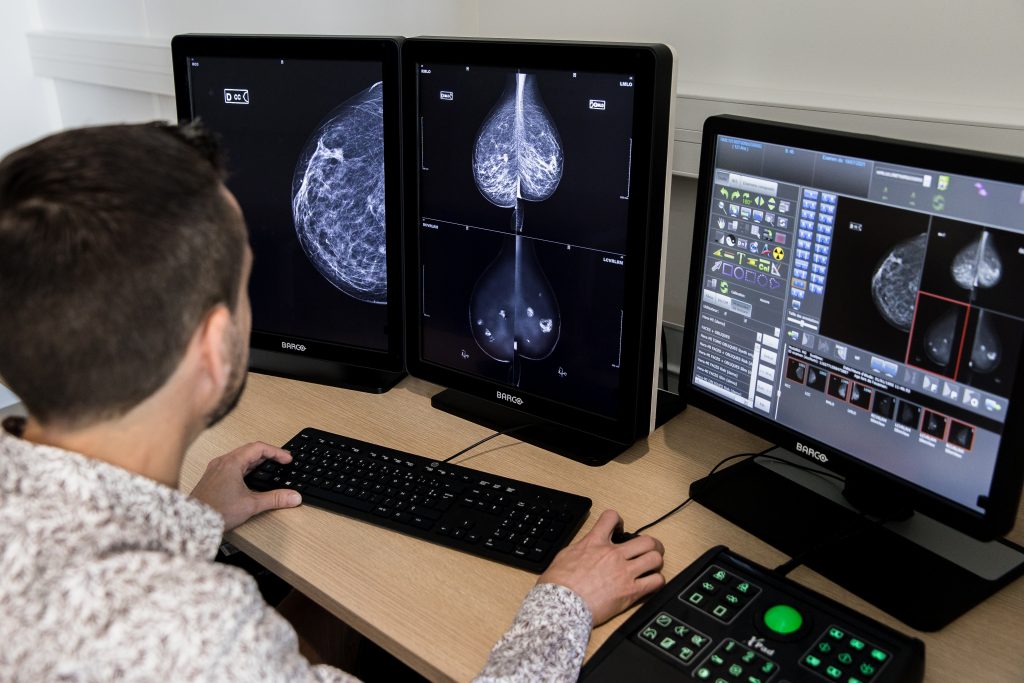
The technical process of the patent is called “negativation” (masking of healthy areas of the breast). Indeed, Breast-SlimView’s algorithm automatically detects and removes all normal physiological areas (vessels, glandular tissue, fatty tissue and mammary gland) and replaces them by artificial fat.
Artificial Intelligence will enable radiologists to decrease the time they spend on non-problematic cases and spend extra time on more complicated ones. Hera-MI helps radiologists to focus on their core business, which is their added value, the analysis of potentially suspicious areas.
In order to offer technological and innovative solutions for breast cancer early detection, Hera-MI wishes to use automated learning techniques and images processing. These solutions improve the diagnosis quality and accuracy while decreasing radiologists’ interpretation time.
Hera-MI’s solutions will optimize breast cancer screening program accessibility. Patients will be treated more quickly, giving them a better chance of being cured of breast cancer.
Based on a technological innovation, Breast-SlimView® includes several innovative features:
- An informative analysis tool for 2D/3D mammography imaging,
- An innovative diagnosis reading support.
CE certified; FDA (in progress)
Breast-SlimView® is a medical device that has been CE certified by the notified body KIWA (0476). It is already marketed in Europe. Hera-MI has also initiated an approval process with the American Competent Authority (FDA) to obtain a marketing certification in the United States.
Hera-MI has an export development strategy, in the United States as well as in the Middle East.
Hera-MI projects:
Nowadays, Hera-Mi’s research is focused on other diagnostic software solutions based on the same operating principle as Breast-SlimView®. The research is focused on cancer, such as lung, prostate, and colorectal cancer.






